In March, with the official expiration of Novo Nordisk's core compound patent for semaglutide in India, this commercially critical GLP-1 molecule entered a state of fully open competition for the first time in a major market, triggering an unprecedented wave of activity across the Indian pharmaceutical industry.
01
Triggering a Collapse of the Pricing System
India is one of the world's largest producers of generic drugs, supplying approximately 20% of the global off-patent drug supply. Major companies have been preparing for this moment for years. Once the patent expires, pharmaceutical companies are no longer bound by the authorization of innovative drug companies and can independently manufacture and sell injectable products containing the same active ingredient. This means India has become the first major country to enter the era of "semaglutide free competition" at the actual market level. According to reports, Novo Nordisk has officially confirmed that its patent protection in India has expired, acknowledging that India's market size, number of diabetic patients, and pharmaceutical manufacturing capabilities make it one of the world's most critical competitive frontiers.
Meanwhile, the high pricing of innovative drugs has long hindered the large-scale adoption of GLP-1 drugs in emerging markets. In India, Novo Nordisk's Ozempic and Wegovy are priced in the range of approximately 8,800 to 16,400 rupees, far beyond the reach of most ordinary patients, which has long constrained market penetration. The patent expiration has created the conditions for a structural breakdown of the pricing system, enabling a larger patient population that previously could not afford innovative treatments to have access to GLP-1 therapy for the first time. With the active ingredient no longer protected, the gap between the core manufacturing cost of the drug and the actual market price has become apparent. India has finally entered a historic period of transformation characterized by "lowering technological barriers, collapsing price barriers, and rapidly releasing market demand."
The most defining feature of this event in India is the historic price collapse—from hundreds of dollars down to just over a dozen dollars. Bloomberg reported that Natco Pharma launched a monthly treatment priced at just 1,290 rupees (approximately $14), which is among the earliest and lowest prices offered by any major pharmaceutical company. Reports from Firstpost and FiercePharma also confirmed that major Indian companies have generally set prices between 1,300 and 5,000 rupees, far below the original pricing system for innovative drugs.
This pricing gap is unprecedented in the history of GLP-1 drugs. For decades, such drugs have been primarily concentrated in Europe, the United States, and some upper-middle-income countries. Due to high R&D costs, complex delivery systems, and strict storage and transportation requirements, they have maintained a high-price system. The maturity of India's generic drug industry has enabled companies to rapidly launch alternative products on the very day of patent expiration, breaking the price monopoly. According to reports, generic versions in India are generally 50% to 80% cheaper than the original drugs sold by Novo Nordisk and Eli Lilly, with some entry-level doses priced as low as one-tenth of the original.
Price reductions not only affect patient behavior but also reshape the entire industry structure. Previously, GLP-1 drugs were mainly concentrated among affluent user groups in first-tier cities. With significant price reductions, patients in second- and third-tier cities and even smaller towns can now enter the market, increasing demand elasticity. As prices fall, demand for GLP-1 drugs shows a highly nonlinear upward trend, which will propel the entire Indian market into a new phase of expansion.
02
Multi-Tiered Competition
The second notable feature of this competition in India is the sheer number of participating companies and the highly complex, multi-tiered nature of the competition. Nearly all major Indian pharmaceutical companies have joined the fray, including Sun Pharma, Dr. Reddy's, Glenmark, Zydus, Lupin, Biocon, Mankind, and others. At least 40 companies and over 50 brands are expected to launch in the short term, forming the most concentrated battlefield for GLP-1 generics in the world.
Table 1: Select Marketed Semaglutide Generics in India
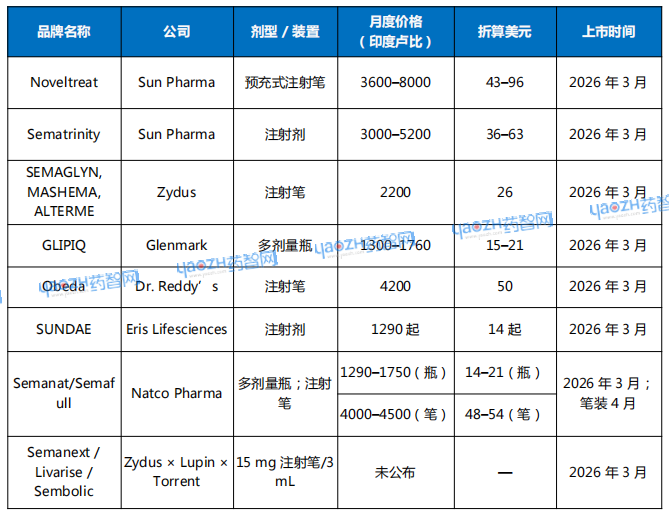
Data source: Bloomberg
Competition is not solely focused on pricing but is also reflected in the diverse innovations in formulation and drug delivery devices. Novo Nordisk's original products use pre-filled injection pens, offering a relatively convenient and standardized dosing experience. However, many Indian companies have adopted different strategies, including multi-dose vials, multi-dose adjustable injection pens, single-use syringes, and glass ampoules with syringes. The availability of different formulations is not only a way for companies to reduce costs but also a commercial strategy to quickly capture market share in the generic drug space. Differences in device experience may become a key factor in patients' long-term choices, because when the active molecule is identical, the injection system becomes the only element that can create differentiation (for injectable formulations).
Changes in channel structure are equally important. During the era of innovative drugs, GLP-1 drugs in India were mainly concentrated in endocrinology and metabolism centers in large cities. Generic drug companies possess stronger capabilities for market penetration, allowing them to reach smaller cities and towns through their extensive sales networks across the country, thereby significantly expanding market coverage. According to Bloomberg reports, many Indian companies have explicitly positioned their market expansion strategies on regions where innovative drug companies have insufficient coverage, leveraging low-price advantages to rapidly establish brand share.
Alliances between companies have further added to the complexity of competition. Collaborations such as Zydus with Lupin and Torrent, market partnerships between Eris and Natco, and joint promotion efforts involving Dr. Reddy's and multiple companies have given competition a group-based, systematic trend. This indicates that companies are considering not just single-brand competition but also more advanced strategic dimensions such as channel positioning and marketing system synergies.
03
Patient ecosystem reconstruction
The launch of generics has not only brought about price reductions but has also triggered a comprehensive rebuilding of the patient management ecosystem. In the fields of weight management and diabetes care, GLP-1 drugs typically involve long treatment durations, with patient compliance, dose adjustment, and lifestyle intervention being critical variables that affect therapeutic outcomes. Therefore, Indian companies have realized that relying solely on low prices cannot establish long-term advantages; they must construct patient management and support systems similar to those of innovative drug companies.
Dr. Reddy's Semakare digital platform is a significant representation of this trend. This system includes a digital application for monitoring weight and lifestyle, offline metabolic centers, and approximately 65 obesity management clinics to be established in the coming months, all aimed at improving long-term treatment adherence, reducing the risk of side effects, and strengthening corporate brand recognition. This model of extending healthcare services has shifted GLP-1 drugs from pure product competition to comprehensive ecosystem competition that integrates medication with value-added management services.
However, the rapid proliferation of generics has also introduced new potential negative consequences. The simultaneous influx of dozens of products into the market may lead to physician confusion, variations in prescribing practices, off-label dosing, and inadequate side effect management. Additionally, while semaglutide is a prescription drug in India, regulatory enforcement is relatively weak, raising the possibility of direct purchases without physician guidance in some urban areas. This could trigger broader misuse or a trend toward cosmetic use. Therefore, as competition intensifies, India may also require stricter regulations to govern the use of this drug.
04
Forecast of Future Market Landscape
India's generic boom is not merely a local phenomenon but a preview of the future evolution of the global GLP-1 market. China and India are experiencing nearly simultaneous patent expirations in 2026, meaning the world's two most significant super-population markets will enter the generic competition phase at the same time. As China has not yet fully opened up generic approval, India has effectively become the first testing ground for real-world competition, with its pricing structure, device innovation, channel penetration, and patient ecosystem development all serving as reference models for other global markets.
This event in India also directly impacts the global strategies of innovative pharmaceutical companies. With the patent cliff clearly approaching, Novo Nordisk has begun implementing pricing strategies and brand protection measures. For instance, according to the Economic Times of India, Novo Nordisk reduced the price of Wegovy by 37% before the patent expiration and established partnerships with multiple companies to maintain channel coverage. However, facing the simultaneous entry of over 40 competitors, innovative drug companies find it difficult to expand market share in India and can only slow the erosion of market share through device differentiation, next-generation products, and more comprehensive patient service systems.
From a global public health perspective, India's low-cost generics may transform the accessibility landscape for obesity and diabetes treatment worldwide. It is estimated that India may have over 440 million overweight or obese individuals in the future, and China faces similar epidemiological trends. Therefore, the widespread availability of low-cost generics will significantly contribute to global public health progress in weight management.
Over the next three to five years, as more countries experience patent expirations, regulatory systems are updated, generic quality standards mature, and supply chains become more diversified, the GLP-1 market is likely to enter a new phase characterized by multipolar competition running parallel with technological evolution. As the first fully open market, India's pricing, channel dynamics, compliance standards, and clinical usage patterns will serve as core reference points for both academia and industry in observing global trends.
05
The Structural Impact of the Chinese Market
As the two most populous countries in the world, both China and India bear a heavy burden of obesity and diabetes, with demand for GLP-1 drugs showing structural growth. Bloomberg has noted that China already had approximately 402 million obese individuals in 2021, with projections suggesting nearly half of its population could be obese by 2050. In comparison, India had 180 million obese individuals during the same period, a number expected to increase to 450 million over the next 25 years. This positions China as one of the most critical potential markets globally after semaglutide's patent expiration in 2026.
The rapid emergence of low-cost generics in India has established a reference point for the world. However, China's regulatory framework ensures that it will not replicate India's "open-market" approach. China emphasizes consistency evaluations, clinical equivalence, pharmacovigilance, and stringent prescription management, which will result in a more stable and controlled rollout of generics, albeit with product quality standards likely exceeding those in India. The extremely low price of $14 in India has already created an expectation effect in the Chinese market, raising public expectations regarding the accessibility and fair pricing of GLP-1 drugs. This will directly prompt innovative drug companies to adjust their pricing strategies in advance. Novo Nordisk's proactive 37% price reduction for Wegovy before the patent expiration in India indicates that companies have entered a preemptive defense phase in global markets, particularly in Asia, and such strategies are highly likely to be implemented in China as well.
More importantly, China is not only a massive consumer market but also a country with globally leading manufacturing and R&D capabilities, possessing all the essential elements to establish an independent system in the GLP-1 arena. Bloomberg emphasizes that China has end-to-end capabilities spanning peptide synthesis, sterile formulation, cold-chain logistics, and the development of complex injection devices. This enables China to not only produce high-quality generics after patent expiration but also to continuously innovate in devices, formulations, and dosage forms. Unlike India's focus on low-price competition, competition in China will place greater emphasis on "quality, technology, and devices."Furthermore, Chinese companies have already formed a rapidly advancing R&D pipeline for GLP-1 and next-generation products, including oral GLP-1s, small-molecule agonists, GLP-1/GIP dual agonists, and triple agonists across multiple proprietary platforms. This indicates that China is not passively waiting for patent expiration but is actively constructing a multi-tiered landscape spanning generics, improved formulations, and original innovation. As domestic companies enter the field, China's GLP-1 market will exhibit a three-pronged model of "high-quality generics + device innovation + original innovation," with competitive quality and technological sophistication surpassing India's price-dominated market structure.
From a public health perspective, the scale of obesity and metabolic diseases in China is far greater than that in India. Therefore, the widespread adoption of GLP-1 drugs will yield profound social benefits. With declining prices and accelerated independent innovation, China has the potential to significantly reduce the disease burden of obesity and type 2 diabetes by 2030, achieving systematic improvements in cardiovascular risk and metabolic complications. This will position China not only as a key consumer market for GLP-1 drugs globally but also as a strategically influential supplier with strengths in quality, innovation, and large-scale supply.The future competitive landscape for GLP-1 drugs will no longer be simply "innovator drugs versus Indian generics," but is more likely to evolve into a multi-centric structure of "Chinese independent innovation + high-quality generics versus the global market," with China occupying a dominant position in the next round of global market reshaping.
06
conclusion
The wave of $14 semaglutide generics in India is not merely a price war, but a structural transformation of the global metabolic disease treatment landscape. The sheer number of companies involved, the rapid pace of device innovation, the depth of channel penetration, and the dramatic decline in prices have all positioned India as a forward-looking window into the future direction of the global market. As the patent cliff advances, the world will witness more markets replicating India's evolutionary path, ultimately shaping a global GLP-1 landscape characterized by lower prices, broader accessibility, and a stronger focus on patient ecosystems.
Ref.
1.Salve,P.et al.India is launching cheap,weight-loss drugs and Novo Nordisk is betting on its brands to stay on top.CNBC.23.03.2026.
2.Sanjay,S.Ozempic Copies at$14 in India as Novo Patent Expires.Bloomberg.20.03.2026.
3.Generic Ozempic to cost Rs 1,290 in India:A game changer in the fight against obesity?Firstpost.20.03.2026.
4.Novo Nordisk patent expiry opens door to cheaper weight-loss drugs in India.CNBC.19.03.2026.
5.Dunleavy,K.With Novo's semaglutide going off patent,Indian drugmakers set to launch their cheaper generics.FiercePharma.20.03.2026.
6.Novo to track generic surge as semaglutide goes off patent.India Times.21.03.2026.