Autoimmunediseases represent the second-largest pharmaceutical market in the world,trailing only oncology, and are a fertile ground for blockbuster drugs.Amongthem, the most representative is the psoriasis drug sector.
Driven bya large patient population, the psoriasis drug sector has given rise to severalblockbuster drugs, including adalimumab, ustekinumab, and risankizumab.Although market competition has become increasingly fierce, the market is farfrom saturated. Instead, it is undergoing a new wave of optimization andupgrading driven by innovative therapies.
01
a fertileground for blockbuster drugs

Throughout thehistory of psoriasis drug development, its essence has been a symbiotic historyof global pharmaceutical companies competing for market share and the iterativeupdating of therapeutic drugs.
The initial treatment for psoriasis was tumor necrosis factor (TNF) inhibitors, which target the immune system by modulating cytokine signaling. A representative drug is AbbVie's Humira (adalimumab): after being approved in 2005 for the treatment of psoriatic arthritis, it subsequently secured indications for plaque psoriasis, pustular psoriasis, and others.
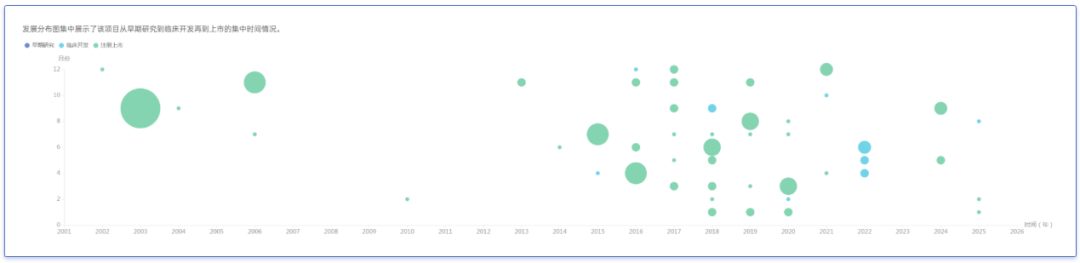
The R&D History of Adalimumab
Source: Yaozhi Data - Global Drug Analysis System
Other TNF-α inhibitors commonly used in global clinical practice have also secured indications for psoriasis, including Johnson & Johnson's golimumab and infliximab, Pfizer/Amgen's etanercept, and UCB's certolizumab pegol.
Thanks to their success in treating autoimmune diseases with large patient populations like psoriasis, humira and etanercept have become blockbuster drugs, each accumulating over $100 billion in global sales since their launch.
There are also several major drugs among interleukin (IL) inhibitors for psoriasis. Representative drugs include Johnson & Johnson's ustekinumab (an IL-12/IL-23 dual inhibitor) and AbbVie's risankizumab (an IL-23 inhibitor). The former reached a peak sales of $10.856 billion in 2023, while the latter achieved global sales of $17.562 billion in 2025 and is expected to become the next "king of autoimmune drugs."
Other IL inhibitors have also performed well. In 2025, Novartis's secukinumab (IL-17A inhibitor), Johnson & Johnson's guselkumab (IL-23 inhibitor), and Eli Lilly's ixekizumab (IL-17A inhibitor) achieved global sales of $6.668 billion, $5.155 billion, and $3.562 billion, respectively.
In addition to biologics, oral small-molecule targeted drugs have become a new treatment option for psoriasis, including PDE4 inhibitors, JAK1-3 inhibitors, and TYK2 inhibitors.
Apremilast is the world's first oral PDE4 inhibitor and the first targeted oral drug approved to treat psoriasis. It was initially developed by Bristol-Myers Squibb's (BMS) subsidiary Celgene and later acquired by Amgen for $13.4 billion. In recent years, apremilast's global sales have remained around $2 billion.
JAK inhibitors have gone through three generations of iteration. Among them, the first-generation tofacitinib and the second-generation upadacitinib have both secured indications for psoriatic arthritis. The third-generation deucravacitinib is the world's first TYK2 allosteric inhibitor and has been approved for the treatment of plaque psoriasis, pustular psoriasis, erythrodermic psoriasis, and active psoriatic arthritis.
In terms of sales performance, Pfizer's tofacitinib reached a peak sales of $2.455 billion in 2021. AbbVie's upadacitinib achieved global sales of $8.304 billion in 2025, a year-on-year increase of 39.1%. BMS's deucravacitinib saw its global sales increase by 18.29% year-on-year to $291 million in 2025.
02
Intense competition phase

Currently, the treatment landscape for psoriasis has become highly diversified, with validated targets such as JAK, IL-17, PDE4, IL-23, and TYK2 becoming hotspots for R&D. The market is currently in a phase of intense competition.

Overview of Psoriasis Indications
Source: Yaozhi Data - Global Drug Analysis System
JAK kinases belong to the intracellular non-receptor tyrosine kinase family, comprising four members: JAK1, JAK2, JAK3, and TYK2. Currently, more than ten JAK inhibitors have been approved globally, and the market is highly competitive.
In the domestic market, foreign pharmaceutical companies such as Pfizer, AbbVie, and BMS have taken the lead in securing the psoriatic arthritis (PsA) indication. Hengrui Medicine's JAK1 inhibitor, ivarmacitinib, is among the frontrunners in China, with a Phase III clinical trial underway for PsA.
In terms of TYK2 inhibitors, BMS's deucravacitinib has already been approved in China for the treatment of plaque psoriasis. However, several domestic companies are conducting Phase III studies, including CSPC Pharmaceutical Group's SYHX1901, Innocare's ICP-488, InventisBio's D-2570, and Hansoh Pharma's HS-10374. Among these, CSPC's SYHX1901 is an oral small-molecule JAK/TYK2 dual-target inhibitor.
Regarding interleukin (IL) inhibitors, the IL-6/12 and IL-17 families have sparked a wave of R&D enthusiasm. The IL-12 family, particularly IL-12 and IL-23, plays a key role in immune-mediated diseases, including plaque psoriasis and inflammatory bowel disease. Meanwhile, the onset and progression of various autoimmune diseases, such as psoriasis and multiple sclerosis, are associated with IL-17.
Currently, several domestic companies have entered the IL-23 targeted drug space, including Innovent Biologics, Akeso Biopharma, China Medical System, Hansoh Pharma, and Qyuns Therapeutics. Among them, Akeso's ebronucimab is the first domestically developed IL-12/IL-23 dual-target monoclonal antibody, approved for the treatment of moderate-to-severe plaque psoriasis (PsO). Innovent's picankibart injection is China's first homegrown IL-23p19 monoclonal antibody, also approved for PsO.
In the IL-36 targeted drug space, only Boehringer Ingelheim's spesolimab has been approved in China for the treatment of generalized pustular psoriasis (GPP). Huaotai Biopharmaceuticals, a subsidiary of Huahai Pharmaceutical, has developed HB0034 (risankizumab biosimilar), which is the first domestically produced IL-36R monoclonal antibody to submit a marketing application.
The PDE4 inhibitor space in China is highly competitive. Amgen/Sun Pharma's apremilast, Pfizer's crisaborole ointment, and Hemay005 (momicrist), the first domestically developed PDE4 inhibitor from Mei Pharma, have already been approved. Numerous domestic companies are following closely, including Hengrui Medicine, Huadong Medicine, China Biopharmaceuticals, Jumpcan Pharmaceutical, and Joincare Pharmaceutical.
It is worth mentioning that in July 2025, Hengrui Medicine licensed its PDE3/4 inhibitor HRS-9821 along with 11 other projects to GlaxoSmithKline (GSK) in a deal potentially worth up to $12 billion. GSK's interest in HRS-9821 is primarily driven by its potential as an add-on maintenance treatment for chronic obstructive pulmonary disease (COPD), independent of prior treatment regimens, with best-in-class potential.
03
The Path to Breakthrough

The treatment of psoriasis has entered a new phase of precision and diversification, with landmark innovative therapies taking center stage.
To fill the revenue gap following the patent expiration of ustekinumab, Johnson & Johnson acquired Icotrokinra (Icotyde), the world's first oral IL-23R antagonist, from Protagonist Therapeutics. Icotrokinra was approved by the FDA in March 2026 for the treatment of moderate-to-severe plaque psoriasis (PsO) in adolescents aged 12 years and older and adults, ushering in the era of oral peptide therapies.
Moreover, Icotrokinra has demonstrated its breakthrough and innovative advantages through a novel mechanism of action, outperforming deucravacitinib in a head-to-head trial for the treatment of PsO patients. Johnson & Johnson projects that Icotrokinra could achieve peak sales of up to $7.5 billion.
UCB's bimekizumab is also a highly potent innovative therapy. As the world's first IL-17A and IL-17F dual-target inhibitor, it has outperformed adalimumab, ustekinumab, secukinumab, and risankizumab in multiple head-to-head studies for psoriasis. Moreover, with approvals for multiple indications including PsO and psoriatic arthritis, bimekizumab achieved global sales of $2.524 billion in 2025.
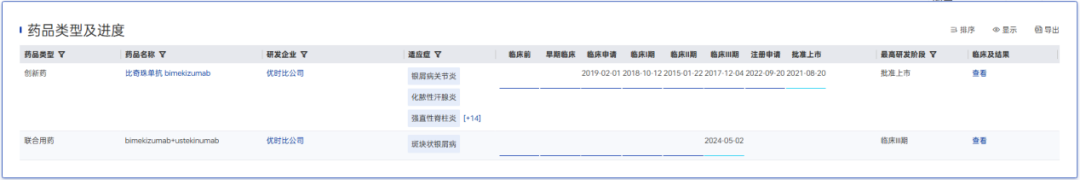
Bimekizumab R&D Progress
Yaozhi Data - Global Drug Analysis System
Although the psoriasis drug market has already become a red ocean, Johnson & Johnson and UCB have both found their paths to breakthrough. These innovative therapies have also attracted domestic pharmaceutical companies in China to follow suit and establish their own pipelines.
For example, Laikanchitamab (LZM012/XKH004), developed by Livzon Pharmaceutical Group in collaboration with Synermore Biologics, has demonstrated multiple advantages including faster onset of action, superior short-term efficacy, long-lasting potent efficacy, high dosing convenience, and excellent safety. In a head-to-head trial for the treatment of PsO, it outperformed secukinumab. Laikanchitamab is currently under regulatory review for PsO, with approval expected by the end of 2026. It is poised to become the world's second and China's first domestically developed IL-17A/F dual-target monoclonal antibody.
In addition, several novel small-molecule targeted drugs are emerging, targeting multiple mechanisms such as A3AR, S1PR1, RORγT, F1FO-ATPase, and RIPK1. Some companies are also exploring the application of PROTAC technology in the autoimmune field.
A3AR is a G protein-coupled adenosine receptor whose expression is upregulated in inflammatory diseases such as psoriasis and rheumatoid arthritis. Currently, the most advanced candidate in clinical development is CF101 (Piclidenoson), licensed by China Medical System from Can-Fite Biopharma. It is currently in Phase III trials for psoriasis and exerts its therapeutic effect by inhibiting the inflammatory cytokines IL-17 and IL-23, thereby inducing apoptosis of pathogenic keratinocytes in the skin lesions of psoriasis patients.
However, the path to exploring novel targets will not be without challenges, and whether these candidates ultimately succeed in becoming approved drugs remains to be validated over time. Nevertheless, it is certain that the psoriasis drug market still holds significant growth potential, and the pathways for innovative breakthroughs remain broad.
04
conclusion

The psoriasis drug field is standing at a critical juncture, connecting the past with the future. The exploration of innovative therapies, novel mechanisms, and new targets is opening up new growth poles for the market.
In this enduring battle of "iteration" and "surpassing," the future winners will be those companies capable of achieving multidimensional breakthroughs in target innovation, clinical efficiency, and commercial capabilities.